Crystallography: one hundred years in the modern era
Crystallography refers to the science dedicated to the study and resolution of crystalline structures. Today, this discipline is present in our daily lives.

Materials science has always shaped, driven, and revolutionized society since the dawn of mankind. It was at the beginning of the 20th century that it began to be studied systematically. For example, it began with the observation that among the great variety of elements and compounds found in nature, many of them have a crystalline structure, i.e. their atoms are structured periodically with a "primordial cell or nucleus" that is repeated over and over again.
This notion is extremely relevant, since knowing the position of an atom in one part of the material and knowing the crystalline structure of the compound allows us to locate the positions of the rest of the atoms of the material and thus determine many of its properties even before performing experiments with them.
Recently, techniques have been developed in laboratories to develop new compounds that, in specialized terminology, grow as crystals. Year after year, new crystalline materials are discovered and/or developed, some of which, when used in state-of-the-art technology devices, from cell phones to space stations, shape, drive, and revolutionize our society once again with such speed that what seemed like science fiction a few decades ago, is now everyday technology, without which it would be impossible for us to perform the most basic tasks.
Today, the technological capabilities of some laboratories even allow us to manipulate two-dimensional crystals, i.e. crystalline membranes only one atom thick, such as graphene. Dubbed the wonder material of the present century, it is a crystalline structure in the shape of a honeycomb, i.e., a hexagon - with carbon atoms at its vertices - that repeats periodically until it forms the membrane of this material. It is estimated that graphene could replace silicon in nanoelectronic devices.
The cell or unit cell is the structure that repeats in all directions of the crystal, specifying the positions of the atoms of the compound. Crystals are then classified according to the symmetry properties of the unit cell. These symmetry properties are also sometimes manifested in macroscopic symmetries of crystals, such as geometric shapes or fracture planes. Thus, a crystalline solid is constructed from the repetition of the unit cell as a function of the lattice parameters, i.e. the lengths of the sides and angles formed by the imaginary lines drawn between the atoms.
Thus, seven crystalline systems can be distinguished: Cubic, Tetragonal, Orthorhombic, Hexagonal, Trigonal, Monoclinic, Triclinic. In the case of two-dimensional crystals, we have only the square, rectangular, hexagonal, and oblique crystalline systems, rectangular, hexagonal, and oblique crystalline systems, with similar characteristics to their three-dimensional counterparts.
Crystallography is the science that is dedicated to the study and resolution of these crystalline structures. This discipline is present in our daily lives. It is not only precious and brilliant geodesies that decorate our rooms or expensive diamonds embedded in rings and necklaces. We find crystals everywhere in our daily lives, for example in table salt, in the development of modern pharmaceuticals, nanotechnology, and biotechnology, and it points to the development of new technology, from toothpaste to liquid crystal displays.
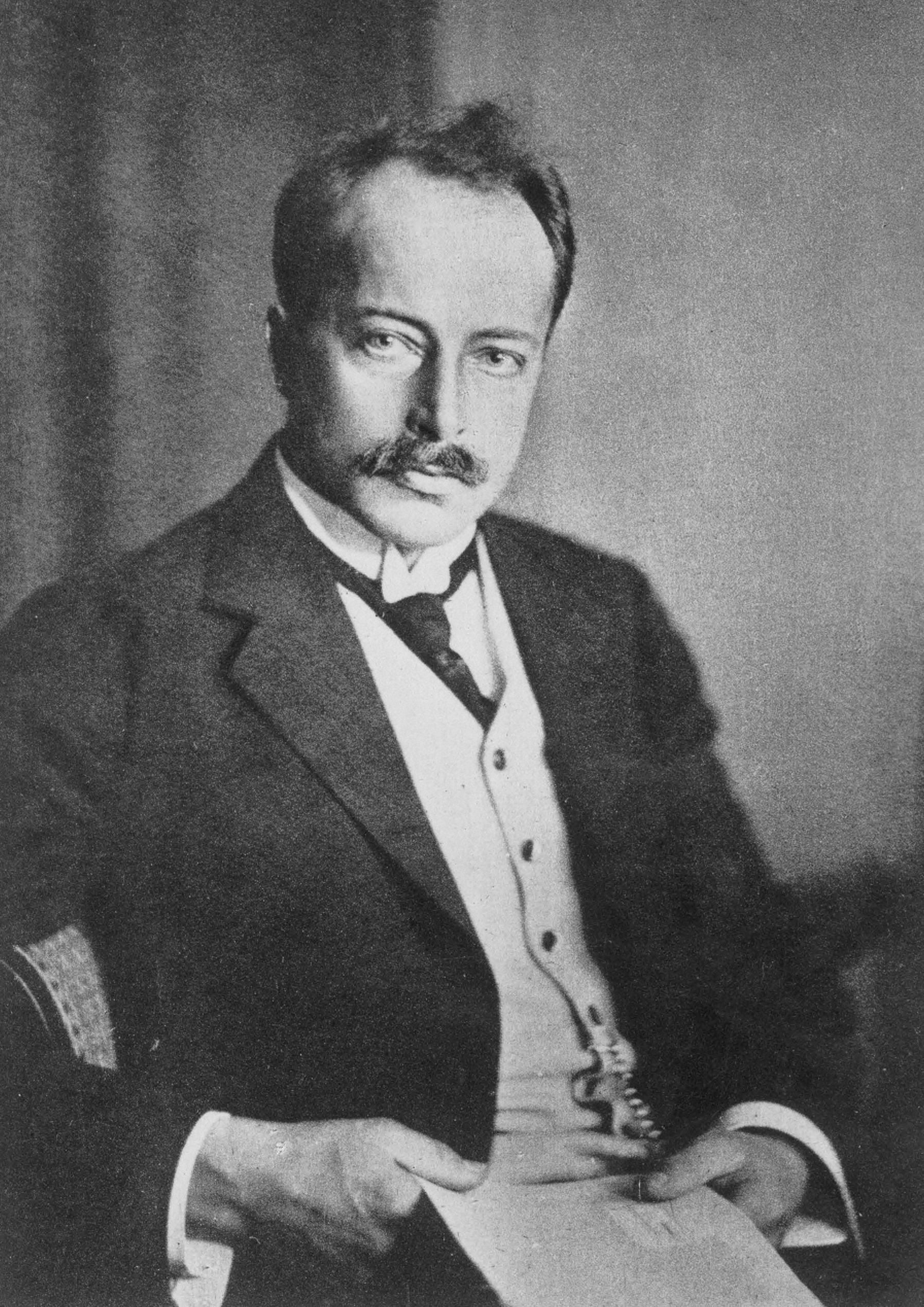
In 1914 Max von Laue was awarded the Nobel Prize in Physics for his discovery of X-ray diffraction through crystals. Thanks to this, he made it possible to better study the structure of crystals (a method called X-ray crystallography).
Written by Alfredo Raya Montaño and Saúl Fernando Hernández Ortiz
Saúl Fernando Hernández Ortiz holds a Master of Science degree in Physics from the Institute of Physics and Mathematics of the Universidad Michoacana de San Nicolás de Hidalgo and is currently a Ph.D. student in Physics at the same institution. He is the author of several articles published in prestigious international journals.
Dr. Alfredo Raya is Professor and Senior Researcher "B" at the Institute of Physics and Mathematics of the Universidad Michoacana de San Nicolás de Hidalgo. He is a member of the National System of Researchers, Level II, Regular Member of the Mexican Academy of Sciences, and Founding Member of the Michoacán Academy of Sciences. He is the author of more than 35 articles in prestigious international journals.
Source: Michoacán University




